Breadcrumb
- Home
- Get Help
- Newsletters
- March 2026 IRB Connection Newsletter
March 2026 IRB Connection Newsletter
Annual Disclosure Due April 30 & New Certificate for Training
Student PI Training Requirement: Reminder to Faculty Advisors and Student PIs
Self-Guided IRB Orientation: New Training for Researchers
Updates to Educational Tools
UI Research Conflict of Interest
All UI faculty, staff and students identified as key personnel who conduct human subjects research are required to complete an “Annual Disclosure of Outside Professional Activities and Interests” via the eCOI online disclosure portal by April 30 each year.
New Training Certificate
For those selecting the “I am a researcher…” when completing their Conflict of Interest (COI) disclosure. The eCOI portal now track the required conflict of interest in research training.
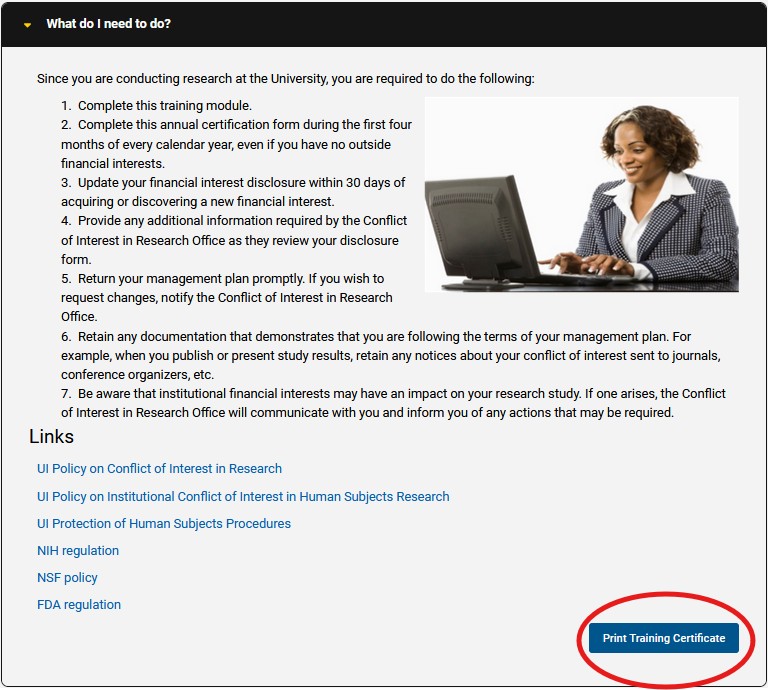
A new feature this year is the ability to print a training certificate upon completion. A training completion certificate was a feature requested by researchers as those who are working with external and commercial IRBs are sometimes required to provide proof of training. The ability to print the certificate is available on the last page of the training module by clicking of the “Print Training Certificate” button in the lower right-hand corner.
If you have already completed training and want a certificate now, or if you need to print a copy of the certificate in the future, follow these steps:
Log into eCOI (https://ecoi.uiowa.edu) with your HawkID and password.
Find your current year disclosure under “Previous Disclosures”.
Click the “View” button.
On the next page, click the “Next Section” button which will take you to the completed training page.
Scroll to the bottom of the page to find the same box (shown in screenshot below) and click “Print Training Certificate” button.
For more details go to the Annual Compliance Requirements webpage.
Student PI Training Requirement: Reminder to Faculty Advisors and Student PIs
Graduate and undergraduate student principal investigators (PIs) are required to view recorded trainings about the HawkIRB New Project form prior to submitting an application in HawkIRB.
Previously, student PIs were required to view a two-part recorded HawkIRB training. The training was recently updated to break the information into 10 shorter sections that allow students to complete the training at their own pace. Each module in the training is 30 minutes or less.
Student PIs must complete the modules in order, view the recordings at regular speed and pass the quiz embedded in each recording and one final quiz to document completion of the Student PI Training Requirement.
The recorded training is posted in the IRB ICON Course for Researchers and consist of the following topics:
1. Good Research Practices-9:36
2. Training Requirements and IRB Submission Requirements-7:50
3. What is and is not HSR and Types of Review-10:05
4. PI, Faculty Advisor, and DEO Responsibilities-27:21
5. HawkIRB Orientation-16:27
6. HawkIRB Navigation-9:28
7. HawkIRB Workflow Communications-5:10
8. HawkIRB Attachments & Edit Functions-8:04
9. Post Approval Responsibilities-21:34
10. Resources and Asking for Help-9:01
In addition to the Student PI Training Requirement, graduate and undergraduate student PIs must also complete CITI human subjects training appropriate for their discipline https://hso.research.uiowa.edu/get-help/icon-course and an eCOI financial disclosure form before HawkIRB will allow them to submit a new project form.
For more information about student PI responsibilities, visit the HSO website.
The Education and Outreach team of the Human Subjects Office (HSO) is happy to announce an update to training that has been in place since 2021. In response to researcher feedback, the training has been updated to help viewers understand what to expect during the IRB review process. This training module contains the same content as the Student PI training requirement; however, this module was designed for researchers to be able to select the specific modules without having to move through them sequentially. The goal of this training is to provide researchers with information about the IRB review process and the HawkIRB system. The training modules are recommended for all new faculty, staff, residents, fellows, professional students, and HawkIRB Delegates who will be using the HawkIRB system.
The new training includes 10 modules that can be reviewed individually. Each module in the training is 30 minutes or less.
The recorded training is posted in the IRB ICON Course for Researchers and consist of the following topics:
1. Good Research Practices-9:36
2. Training Requirements and IRB Submission Requirements-7:50
3. What is and is not HSR and Types of Review-10:05
4. PI, Faculty Advisor, and DEO Responsibilities-27:21
5. HawkIRB Orientation-16:27
6. HawkIRB Navigation-9:28
7. HawkIRB Workflow Communications-5:10
8. HawkIRB Attachments & Edit Functions-8:04
9. Post Approval Responsibilities-21:34
10. Resources and Asking for Help-9:01
For more information about human subjects research training, visit the HSO website.
Recent updates to HawkIRB Section VII and a new Part 11 Compliant electronic consent (eConsent) have necessitated updates to four educational tools provided on the Human Subjects Office (HSO) website.
The updates include new versions of the following educational tools related to Part 11 Complaint eConsent:
Additional updates have been made due to recent changes in HawkIRB Section VII:
Educational tools are provided by the HSO to offer supplemental guidance regarding regulations, policies and best practices for study procedures when completing submissions for IRB review. For more information visit the Educational Tools webpage.