Breadcrumb
- Home
- Get Help
- Newsletters
- July 2025 IRB Connection Newsletter
July 2025 IRB Connection Newsletter
AAHRPP Reaccreditation
Genetic Testing in Human Subjects Research
Herky Hint for HawkIRB: Genetic Testing
HawkIRB Informed Consent Template Language for Genetic Research
The Human Subjects Office (HSO) is pleased to announce the University of Iowa Human Research Protections Program (HRPP) achieved reaccreditation through the Association for the Accreditation of Human Research Protection Programs, Inc (AAHRPP) on March 15, 2025. The reaccreditation is good for the next five years.
AAHRPP accreditation is an international industry standard reflecting a commitment to following the highest standards of ethics, quality, and human subjects protections. At the University of Iowa, this deep commitment is only possible with support from institutional leadership, IRB membership, HSO staff, and the greater research community. The University of Iowa HRPP is proud to have been the first academic institution to achieve AAHRPP accreditation in 2003. This year’s reaccreditation is the fifth reaccreditation for the UI program
The reaccreditation process is a two-step, yearlong process. The first step is a review of the HRPP policies and standard operating procedures, and the second step is a two- to three-day site visit. The site visit, which took place virtually in October of 2024, involved interviews with over 70 individuals from the HSO, IRB members, HRPP members, and the research community.
The UI HRPP was recognized for the following strengths as part of the reaccreditation process:
The Office of Vice President for Research and the Human Subjects Office launch of the “IRB Efficiency Initiative,” a multiyear quality improvement project initiated utilizing an external consultant to be followed by a multiphase implementation with improvements targeted at the electronic IRB system, IRB operations, turnaround times, transparency with research teams, and educational tools. (Standard I-2 and Element I.5.B.)
The Human Subjects Office and the Institute for Clinical and Translational Science offer a wide range of educational sessions, including the Clinical Trials Bootcamp for faculty investigators, new employee onboarding, monthly lecture series, and the Research Academy for research team members. Researchers indicated the programs helped their staff improve their knowledge and skills. (Element I.1.E.)
In particular, HSO leadership would like to thank the HSO staff, the IRB members, and the HRPP Committees for all their hard work preparing for the AAHRPP reaccreditation. The success of the HRPP program is not possible without all the hard work and dedication from each and every one of you. Thank you.
Genetic testing in research involves scientists examining a person's saliva, blood, or cheek swab to study DNA, RNA, mitochondrial DNA, chromosomes, or proteins. The purpose of the research might be to find out:
• Why some people get certain diseases
• Whether a person can pass a disease to their children or inherit a disease from their parent
• How a person’s genetic makeup may affect how react to different medicines
There are some kinds of research that include DNA or RNA tests that are not considered genetic testing because they do not look for inherited traits or changes in a person's genes. For example, scientists who study the gut biome may look at the DNA of bacteria living in the body, but because the analysis does not involve looking at an individual’s DNA, it is not genetic testing. Similarly, some infectious disease testing looks at the DNA or RNA of viruses or bacteria (like COVID-19 or HIV). Or some cancer researchers extract DNA from tumors to find mutations. If these mutations only exist in the tumor, it is not considered genetic testing of the person.
What is Unique about Genetic Research?
Although most aspects of reviewing research that includes genetic testing are familiar human subjects research protections, genetic testing includes some unique aspects that must be taken into consideration:
The research on the specimen may never end.
The ability to withdraw data/specimens is limited.
Confidentiality risks may persist in perpetuity.
The participant may have difficulty understanding exactly what is being studied.
Results provided back to participants may negatively impact their lives and the lives of their families.
UI Policies and Procedures
The UI Standard Operating Procedures and Researcher Guide Section II, Part 10 outlines a number of specific requirements involving genetic research. It is important for researchers to be familiar with the information and provide the following:
General information about DNA, and specific information being collected for the study.
The extent of subject and sample confidentiality if the sample and subsequent information will be part of a registry or database
The rights and limitations of subjects to require destruction of their sample and/or associated data at a future date. The rights and limitations of subjects to require that their sample and or associated data be stripped of any identifying information.
If identifying information will be available to other researchers if their sample and/or associated data are part of a registry or database (required for all studies, regardless of being a registry or database)
An explanation of the mechanisms for maintaining confidentiality in long-term studies, registries, or databases.
Potential for commercial profit by the institution, investigator, or sponsor from information gathered in this study.
The access to genetic counseling in cases where a study may reveal genetically important information (i.e., possessing genetic defects which could be passed on).
Subjects must have the right to decline receiving genetic information
In the absence of a specific authorization to maintain a DNA sample beyond the initial project, DNA samples collected, stored, and/or analyzed in connection with a research project must be destroyed upon completion of the project or withdrawal of the individual from the project. This information must be clearly stated in the Informed Consent Document
Other Regulatory Requirements Related to Genetic Testing
GINA (Genetic Information Nondiscrimination Act) protects individuals from genetic discrimination by health insurers and group health plans, ensuring genetic data cannot influence coverage or premium decisions. However, it excludes protections for life insurance, disability insurance, long-term care insurance, and manifest genetic conditions, while allowing subjects to opt out of genetic research for privacy concerns.
CLIA (Clinical Laboratory Improvement Amendments of 1988) ensures appropriate laboratory testing standards are in place for the sharing of genetic testing results.
Conclusion
Understanding the unique requirements of genetic testing allows the IRB to assist the PI in providing comprehensive information in HawkIRB and the informed consent document in order to better protect human participants in genetic research.
For information about the relevant sections of HawkIRB, and the informed consent template language, see this month’s Herky Hint.)
Herky Hint for HawkIRB: Genetic Testing
When submitting a HawkIRB application for a research project that includes genetic testing, the principal investigator will need to ensure that the HawkIRB application and the informed consent documents explain:
What type of tests will be conducted with the blood, tissue, or specimens?
What will happen to the blood, tissue, or specimens as relates to results, storage, and sharing?
The details that go into these two categories are complex, and these complexities can lead to inconsistencies within the HawkIRB application and between the application and the informed consent document. Inconsistencies are likely to increase IRB review time and slow approval.
HawkIRB Sections VII.C.1 – VII.C.12 Genetic Testing
To avoid inconsistencies, a study team should complete the HawkIRB application before the informed consent document, because responses to certain questions will help build templated language into the informed consent document automatically.
Section VII.C in HawkIRB is where the researcher will provide most of the information about genetic research. Responses should be in lay language so any non-scientific HSO\IRB member can understand what is happening.
VII.C.1 Does this project involve any research on genes or genetic testing/research?
Should be marked ‘Yes’ if specimens will be collected during the study for the purposes of genetic testing that will or may be conducted under this protocol. This includes plans for genetic testing on these samples during this study or sometime in the future, after the sample collection and interventions (if any) have been completed. When the PI selects ‘Yes’ to this question, template language is inserted into the consent document to describe genetic research to subjects. Additionally, when VII.C.1 is answered “Yes,” the UI informed consent template sections are required:
“Genetic Information Nondiscrimination Act (GINA)” and
The genetic risk addition in the “WHAT ARE THE RISKS OF THIS STUDY?”
“Genetic Research”
“Tissue/Blood/Data Storage for Future Use,”
In the “Tissue/Blood/Data Storage for Future Use” section, the researcher must outline the study specific confidentiality protections and indicate how they will store genetic specimens and data. (The UI informed consent template also recommends either indicating if the collection is mandatory or optional, allowing the subjects the choice for future use.)
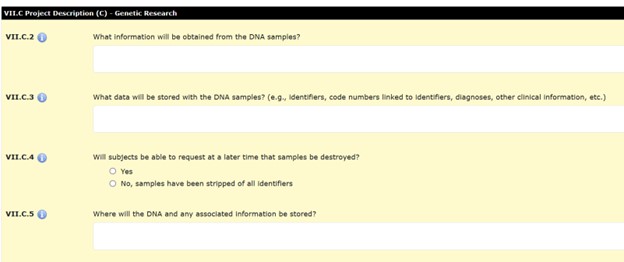
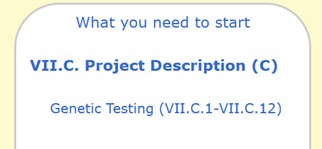
VII.C.2 What information will be obtained from the DNA samples?
The response should include all DNA or genetic tests and the data that will be subsequently collected as a result of the testing of the DNA samples.
VII.C.3 What data will be stored with the DNA samples? (e.g., identifiers, code numbers linked to identifiers, diagnoses, other clinical information, etc.)
Describe what data points may be physically stored with the sample. Details here should be described in the confidentiality section of the consent.
VII.C.4 Will subjects be able to request at a later time that samples are destroyed?
If the samples are stripped of all identifiers, subjects will not be able to request their destruction at a later time. This information must also be in the consent document.
VII.C.5 Where will the DNA and any associated information be stored?
The response to this question should provide detailed information about the location(s) where the samples AND any associated information will be shipped, shared, and/or stored. The information here should be consistent with VII.C.7, X.5 and XII.

VII.C.6 Describe the mechanisms for maintaining confidentiality at the storage location.
The information provided must specify where specimens will be kept, and may include a locked freezer or storage unit, locked lab, and other security protections at the storage location. This information must also be in the consent.
VII.C.7 Could the DNA and/or associated information be shared in the future with other researchers?
“Other researchers” includes researchers who are not part of the University of Iowa research team identified as part of this project in HawkIRB. This could include collaborators outside the University of Iowa or UI researchers who are not named on the research team. This information must also be in the consent. If ‘Yes’ is answered, VII.C.8 will open, asking for a description of the sharing procedure.
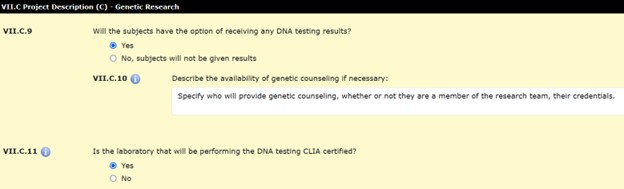
VII.C.9 Will the subjects have the option of receiving any DNA testing results?
If ‘Yes’ is selected, VII.C.10 will require a response. The study team is not required to share the results of genetic tests. Whatever approach the study team makes must be described in the IRB application and the informed consent document.
VII.C.10 asks for a description of the availability of genetic counseling. Use of a genetic counselor is required if the specific research related genetic testing results will be shared with subjects. The response should specify who will provide genetic counseling, what their credentials are, and whether or not they are a member of the research team. The individual(s) providing genetic counseling must have appropriate credentialing to provide this type of counseling. The study team should also consider whether or not participants will be able to receive or opt out of receiving test results. (Note: This must be clearly included in the consent.)
The study team can choose not to share the specific results of genetic testing. If the team makes this decision and identifies an unanticipated outcome, the study team can state in the informed consent document if an unexpected outcome is identified the study team will refer the subject to their clinical care team for clinical testing. (The clinical testing would not be considered part of the research study.)
VII.C.11 Is the laboratory that will be performing the DNA testing CLIA certified?
If the response to VII.C.9. is “Yes,” the response to VII.C.11 must also be “Yes.” If research results of a genetic test are given to subjects or placed in the medical record, the genetic test must be performed in a CLIA-certified lab.
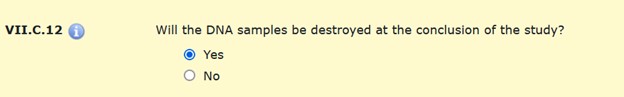
VII.C.12 Will the DNA samples be destroyed at the conclusion of the study?
Unless the study is approved to store samples for future research. (See HawkIRB Section VII.C.7 and XII.C.4.) samples MUST be destroyed when the study is closed through HawkIRB. The destruction of samples must also be included in the consent document.
Other HawkIRB Sections
Remember that although one section (VII.C.) holds the questions specific to genetic testing, there are other sections where the PI may provide information that overlaps with or supports responses in VII.C.
For example, Question VII.E.6 ask for a detailed description of the study procedures.

Question X.4 asks how information/data and samples will be collected and stored.
Section XII addresses future research.

In order to avoid inconsistencies, the PI must carefully review the information provided in these sections to make sure it is consistent with the information provided in section VII.C. and the consent document.
For more information about genetic research see this month’s IRB Connection newsletter article, and the related article about the informed consent language.
HawkIRB Informed Consent Template Language for Genetic Research
The informed consent template provided in HawkIRB includes a number of different sections that address information required when genetic testing may occur as a condition of the research. It is important for researchers to understand each of the sections and know when they should be included in the informed consent document.
The information provided in the informed consent document and the IRB application must be consistent and accurately reflect what will happen to subjects during the course of the research. The informed consent checklist is available to help researchers correlate sections of the HawkIRB application with the informed consent document.
This article summarizes the standard informed consent template language. The informed consent template language includes bold or bracketed statements that are either instructions for the study team or language that can be customized to the relevant study procedures.
Tissue/Blood/Data Storage for Future Use
The following conditions must be explicitly stated in this section of the informed consent document. The research team must carefully consider whether:
It will\will not be possible for future research use of tissue/blood/data to be performed.
Whether or not the use of tissue/blood/data will\will not include whole genome sequencing as part of the future use.
Whether or not the tissue/blood/data will be stored with\without any identifiers or if it will be completely de identified.
Blood cells removed from the blood samples will be used to make a cell line and DNA or conduct whole genome sequencing. Cell lines are produced by growing blood cells in a laboratory and allow us to have a source of DNA without having to redraw your blood. These blood cells can be stored for decades or more. The [cell lines, DNA, and\or genome sequencing results] and data will be made available to researchers trying to learn more about the cause of diseases.
Each of the cells in your body contains DNA. DNA is the instruction manual that determines your appearance in things like eye color or how tall you can be. Your DNA may also lead to a higher or lower risk of certain diseases. Your environment will also determine some of your disease risks.
Your DNA is a string of four building blocks, called “bases.” These bases are represented by the letters G, A, T, and C. There are billions of these letters strung together in every human’s DNA and they are arranged in packages like words. Each of these “words” have specific jobs in the body. Most of the time the letters are the same in everyone. But about 1% of the population might have an “A” where someone else has a “G.” This difference can explain why some people have blue eyes and others brown eyes, or why some have a high risk for a certain cancer and others a low risk. All these letters come together to create your “genome sequence,” a kind of book of your genetics. It now possible to read off each of these letters and read your complete genome sequence. Your DNA sequence is unique to you. You inherit your DNA in almost equal parts from each of your parents. In very rare cases, your genome can also change through “mutations.” A mutation is like if you tried to copy a page from a book but misspelled some words. Mutations usually result from copying errors that occur in certain letters when being passed from parent to child.
When we take a sample of your blood/tissue for this study, it will go to a lab to read off those letters and give us a report on your genome. This information will then be compared with other genomes to see how they may be the same or different. It is our hope this will help us to better understand how the human body works and/or what causes it to not work well, as when someone has a disease.
The tests we might want to use to study your [type of tissue/blood/data] may not even exist at this time. Therefore, we are asking for your permission to store your [type of tissue/blood/data] so that we can study them in the future. These future studies may provide additional information that will be helpful in understanding [disease/condition], but it is unlikely that what we learn from these studies will have a direct benefit to you. It is possible that your [type of tissue/blood/data] might be used to develop products tests, or discoveries that could be patented and licensed. In some instances, these may have potential commercial value and may be developed by the Investigators, University of Iowa, commercial companies, organizations funding this research, or others that may not be working directly with this research team. However, donors of [type of tissue/blood/data] do not retain any property rights to the materials. Therefore, there are no plans to provide financial compensation to you should this occur.
[Select one of the below paragraphs to reflect how the tissue/blood/data collected as part of the study will be identified\deidentified once it is collected.]
Your [type of tissue/blood/data] will be stored with a code which may be linked to [insert what identifiers may be associated with the code (e.g. your name or any other kind of link that would enable us to identify which sample(s) are yours, DOB, etc.)]. If you agree now to future use of your [type of tissue/blood/data] but decide in the future that you would like to have it removed from future research, you should contact [name and phone number of PI]. However, if some research with your [type of tissue/blood/data] has already been completed, the information from that research may still be used.
--- OR ---
Your [type of tissue/blood/data] will be stored without your name or any other kind of link that would enable us to identify which sample(s) are yours. Therefore, if you give permission to store your [type of tissue/blood/data], it will be available for use in future research studies indefinitely and cannot be removed.
[If subjects can participate in the main study without giving permission for future use of tissue/blood/data, consider using “yes/no” check boxes for the subject to indicate permission for the optional future use.
Examples of such “yes/no” options are given below: You may revise these to apply to your study.] Please place your initials in the blank next to Yes or No for each of the questions below:
My [insert type [type of tissue/blood/data] ] may be stored/shared for future gene research in (e.g. cancer, heart disease, etc.)
_____ Yes ____ No
My [insert type of [type of tissue/blood/data]] may be stored/shared for future research for any other purpose.
_____ Yes ____No
Will I be notified if my [Data\biospecimens\images] result(s) in an an unexpected finding?
… you will not be informed of any unexpected findings. The results of your [data/biospecimens/images] will not be placed in your medical record with your primary care physician or otherwise. OR
We may learn things about you from the study activities which could be important to your health or to your treatment. If this happens, you can decide whether you want this information to be provided to you…
Genetic Research
One purpose of this study is to look at genes (DNA) and how they affect health and disease. Genes are the instruction manual for the body. The genes you get from your parents decide what you look like and how your body behaves. They can also tell us a person’s risk for certain diseases and how they will respond to treatment.
You are being asked to give a [insert type of sample, e.g., blood, urine, etc.] for genetic research. What we learn about you from this sample will not be put in your health record. [Note to investigators: Results of a genetic test may be given to subjects or placed in the medical record only if the test is performed in a CLIA-certified lab.] [If applicable, insert: Your test results will not be shared with you or your doctor.] No one else (like a relative, boss, or insurance company) will be given your test results.
[Results of a genetic test may be given to subjects or placed in the medical record only if the test is performed in a CLIA-certified lab. If receiving the results of the genetic test is optional, include a yes/no check box for subjects to indicate their choice. Also include whether or not genetic counseling would be available and who would pay for such counseling.]
Genetic Information Nondiscrimination Act (GINA)
A federal law called the Genetic Information Nondiscrimination Act (GINA) generally makes it illegal for health insurance companies, group health plans, and employers of 15 or more persons to discriminate against you based on your genetic information. Based on this new law, health insurance companies and group health plans are prohibited from requesting your genetic information that we get from this research. This means that they may not use your genetic information when making decisions regarding your eligibility for insurance coverage or the amount of your insurance premiums. Be aware that this new federal law will not protect you against genetic discrimination by companies that sell life insurance, disability insurance, or long-term care insurance. The law also does not prohibit discrimination if you are already known to have a genetic disease or disorder.
What are the risks of this study?
Genetic Research
One risk of giving samples for this research may be the release of your name that could link you to the stored samples and/or the results of the tests run on your samples. To prevent this, these samples will be given a code. Only the study staff will know the code. The name that belongs to the code will be kept in a locked file or in a computer with a password. Only [investigator’s name and/or other’s names] will have access to your name.
For more information about genetic research see this month’s IRB Connection newsletter article and the associated Herky Hint.
