Breadcrumb
- Home
- Get Help
- Newsletters
- March 2025 IRB Connection Newsletter
March 2025 IRB Connection Newsletter
Participant Recruitment
Herky Hints for HawkIRB: Subject Recruitment
Upcoming Changes to the UI Ethics in Research Policy
When planning for recruitment of research participants, the principal investigator (PI) and the research team need to consider how they will communicate with their intended participant population. The IRB, regardless of whether it is a local UI IRB or an external IRB, must review and approve all recruitment methods and materials prior to their use.
Guidance and Best Practices
The UI Standard Operating Procedures and Researcher Guide provides information in section II. 11.A.ii Recruitment materials & methods.
The recruitment message is an invitation for people to consider participating in the research and is the beginning of the consent process. Messages should be brief and informative and written in plain language. A research study may need different recruitment messages for different subject populations.
It is good practice to plan more than one recruitment method at the beginning of the study and to describe each of them in the HawkIRB application and/or the external IRB application. The research team does not need to use all of the IRB-approved recruitment methods but including them is helpful in case the primary method is not as effective as expected.
Note: the application for commercial/external IRBs only has two options for recruitment methods. Otherwise, the applications and processes are the same.
A Recruitment Letter Template, available on the HSO website, includes standard language and prompts to include study-specific information to assist researchers with preparing recruitment letters and email messages. The HSO strongly encourages the use of this template as it has been endorsed by the IRB, UI Health Care Joint Office for Compliance, and Human Research Protection leadership.
Recruitment messages should:
State the purpose and/or a brief statement of what is expected of the subject
Include the time commitment for study participants
Indicate the department affiliation of the Principal Investigator and where the research will take place
Provide the name and contact information for a contact person on the research team
Summarize the study inclusion and exclusion criteria to help potential subjects self-screen
Researchers may state the compensation amount in recruitment materials. However, the language or graphical elements for recruitment materials should not emphasize compensation or present study information in a way that could be considered coercive to potential subjects. Additionally, the language and graphics used should not be perceived as “marketing” the study.
Recruitment messages should not:
1) Emphasize the payment amount.
2) Include the name of commercial sponsors or products.
3) Use phrases such as "help needed" or "subjects wanted.” Instead use "you are invited"
or "participants invited."
4) State or imply a certainty of a favorable outcome or other benefits beyond what is
outlined in the consent document and protocol.
5) Emphasize how “important” the study is. Nor should it include any exculpatory language.
6) Make exaggerated marketing claims or sales pitches
7) Make claims about the safety or efficacy of the study drug or device or include statements that are inconsistent with Food and Drug Administration (FDA) labeling
8) Use terms such as “new treatment,” “new medication,” or “new drug,” without stating that the medication is investigational.
9) Promise free medical treatment when it is just that participants will not be charged for taking part in the study
The Researcher Guide includes guidance about certain recruitment methods that are not allowed, including cold-calling and the use of finders' fees.
The Guide also provides information about:
Use of protected health information in the medical record for recruitment purposes, also known as a partial waiver of HIPAA authorization (section II, part 11.A.i)
Use of UI mass email for research recruitment (section II, part 11.A.ii.b)
Research Match.org (section II, part 11.A.iv)
Additional Considerations
IRB approval stamp - Some recruitment outlets may have their own specific guidelines for format and content and may require documentation of IRB approval. For example, while the IRB does not require an approval stamp on recruitment messaging, an IRB approval stamp is required for Noon News announcements, Cambus posters and press releases distributed through UI channels. It is important to consult the guidelines for each office or department prior to the IRB-submission of the advertisement, poster or message.
Positions of power or authority - When the investigator or a member of the research team is in a position of authority over potential subjects (such as an instructor recruiting students in their class or a health care provider recruiting their own patients), to minimize the possibility of coercion or undue influence it may be best for someone else on the research team to conduct the recruitment and consent process and to be the contact person on the research team.
Use of educational records - Under the Family Educational Rights and Privacy Act (FERPA), recruitment activities can only be conducted by the instructor and others who are authorized to have access to educational records (including who is enrolled in the course). For additional information, see the educational tool Research Involving University of Iowa Student Records.
IRB Approval
All aspects of the recruitment plan and content must be reviewed and approved by the IRB before participant recruitment can begin. Recruitment materials (including posters, emails, videos, letters, etc.) reviewed by the IRB will be reviewed as the final product. Therefore, it is important that they contain all information, text, and graphic design elements. If videos are used as a subject recruitment tool, provide either the URL if the video is posted online or upload it to HawkIRB. If the video file is too large, the PI can create a OneDrive file. Add the location of the file in the IRB application and share it with the HSO staff and IRB Chairs involved in the review.
Once the recruitment plan and materials have been approved by the IRB, no changes to the methods or materials may be made without having the changes approved by the IRB by submitting a modification form in HawkIRB. There may be an exception to this requirement for research that qualifies for exempt status. See the UI IRB Standard Operating Procedures and Researcher Guide, Section I, Part 13 (Exempt Human Subjects Research – Modifications to an Exempt Application).
For more information about recruitment planning and materials, there are two recorded lectures in the IRB ICON Course for Researchers, under Additional Topics:
A Deeper Dive into the Research Process – Recruitment Process
Make Effective Recruitment Plans
For more information about submitting recruitment plans and materials for approval, see the Herky Hint for HawkIRB in this month’s newsletter.
By Emily Shultz, CIP
When describing study recruitment in HawkIRB, there are several layers of information that must be provided in section VII.D, and in the attachments section(s).
Methods
Section VII.D.1 must include all potential methods that are likely to be used to communicate information about the study. While not all the recruitment methods you indicate have to be implemented, it is wise to include methods you may consider optional. For example, if you don’t indicate email as an option in VII.D.1., then you can’t use email for recruitment unless you submit a modification form for IRB approval. It is also important to remember: all methods checked here should also be described in section(s) VII.D.29 and/or VII.D.30.
When certain recruitment methods are selected in section VII.D.1, an attachment field will appear to upload the relevant materials. For example, if the study will use the UI mass email system as a recruitment method, provide a copy of the email content in the appropriate attachment category. The same is true for advertisements, posters, brochures, letters, and news releases.
If a website will be used for recruitment, provide the URL in the open text field. This method does not trigger an attachment category to open. Attach screen shots or a Word document with the website content in the Miscellaneous Attachments category of the attachments page.
For recent changes to the UI mass email process, see also: Upgrades To Research Recruitment Emails in 2025.

If an existing registry or database will be used as a source for contacting potential subjects, provide information about who controls it, how individuals are entered into it, the process of obtaining information from the pool, and who authorized access to the registry/database. If the database is from a previous or ongoing research project or registry, also include the IRB ID#.
Note: Commercial/External IRB applications only have two options for recruitment: email or researchmatch.org. Other areas remain the same for both local and external IRB applications.
If the study will use medical information or clinic records from UI Health Care, its hybrid entities or the Iowa City VA Health Care System (VAHCS) as the method to identify potential subjects for research purposes, the response to question VII.D.1.a. must be “yes.”

If you are accessing patient or clinic records without authorization of the individual, include justification that your work meets the requirements for a partial waiver of HIPAA authorization in section VII.D.2-7.
For more information about requesting a partial HIPAA waiver, see the UI Standard Operating Procedures and Researcher Guide Section II.11.A.i “Partial Waiver of HIPAA Authorization and a waiver of consent for recruitment purpose.”
Contact Prior to Consent
Sections VII.D.8 – 11 are related to discussing the study with subjects prior to consent, either in person or by phone. These questions refer to both the recruitment and consent process; interactions that happen prior to enrollment.
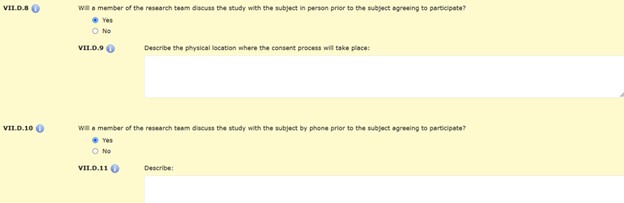
Sections VII.D.12 through VII.D.18. are specific to the consent process. Additional information about participant screening and obtaining consent are requested in sections VII.D.19 through VII.D.28.
Participant Enrollment
Sections VII.D.29. and VII. D. 30. include prompts for a great deal of information about the recruitment and enrollment or consent process.
Provide details about how and when recruitment materials will be used. Refer by name to document(s) on the attachments page, as necessary. Provide a detailed description for the use of all recruitment materials listed in Section VII.D.1.
Describe the recruitment and consent process for adults in section VII.D.29, and for children in section VII. D. 30.


Attachments
Attach recruitment materials in the appropriate attachment categories. Although the IRB does not require an IRB approval stamp on recruitment materials, researchers can use the blank stamped template to include a stamp placeholder. This template is available in the drop-down menu in the Consent/Assent category and in the individual attachment categories for specific types of recruitment materials. The HawkIRB system can only populate the stamp in Word (.doc) and rich text format (.rtf) documents. It may not be possible to include a stamp on a pdf or PowerPoint poster. The IRB can only provide a stamp on documents in the recruitment and consent attachment categories; not in any other attachment category.
More details about the recruitment process are available in the UI Standard Operating Procedures and Researcher Guide Section II, part 11: HawkIRB Section VII.D. Recruitment & Consent Process.
Questions about this topic? Email the HSO Education and Outreach team or come to Office Hours to speak directly to a member of the HSO team.
Have an idea for a future Herky Hint for HawkIRB? Let us know!
Upcoming Changes to the UI Ethics in Research Policy
By Mike Andrews, Director of Research Integrity and Security
The Research Integrity and Security Office (RISO), a compliance unit under the Office of the Vice President for Research (OVPR), is working to update the University of Iowa’s (UI) Ethics in Research Policy (Chapter II §27.6 of the UI Policy Manual) and procedures for research misconduct. The changes are, in part, necessitated by the release of the Final Rule for the Notice of Public Rule Making (NPRM) to 42 CFR §93.
The NPRM for 42 CFR §93 was finalized In September of 2024. Academic institutions, such as the UI, are required to update and implement the changes in the regulation into their policies and procedures by January 1, 2026.
The UI’s Ethics in Research Policy was last updated in 2014. The policy is very PHS-focused and does not reflect differences in how other federal agencies process cases of alleged research misconduct. The new institutional policy will be less focused on the Public Health Service (PHS)-guidelines and be responsive to policies from other federal agencies and non-federal sources of funding. The draft of the new Ethics in research policy
updates the new language from the Final Rule,
includes new language for the assessment phase,
new and updated definitions, and
changes to the inquiry and investigation timelines.
In order to make the policy more flexible, we have pulled many of the PHS-specific procedure elements out of the policy and created a new PHS-specific research misconduct procedure document. The plan is to create similar procedure documents for other federal funding agencies (e.g., the National Science Foundation, the Department of Defense), as well as creating a procedure document for cases that do not involve federally funded work.
The suggested changes to the policy are in the review process which includes review by the Vice President for Research (VPR), Office of General Counsel (OGC), the Associate Provost for Faculty (APF), the Research Council, and the Faculty Senate. The research misconduct procedures documents will also be reviewed by the by the VPR, APF and OGC.
The Final Rule for 42 CFR §93 has many changes incorporated into it, including:
updated language for time limitations to initiate research misconduct proceedings and provided clarification of the “subsequent use” rule,
new language for instances where a respondent either intentionally destroys research records after being notified of a research misconduct proceeding or refuses to cooperate and provide records. Destruction of records is now considered knowingly or intentionally committing research misconduct,
new language to the confidentiality section on who may have a need to know regarding information related to a research misconduct proceeding, and states that the confidentiality requirement no longer applies once a final determination has been made,
clarification on some existing definitions and addition of a number of new definitions,
guidance on institutional sequestration procedures and new language for how institutions handle cases involving multiple respondents and/or multiple institutions,
new language adding assessment as a formal phase of a research misconduct proceeding,
addition of new language to the institutional process for conducting inquiries, and changing the timeline for completion of the inquiry phase from 60 to 90 days,
addition of new language for procedures to conduct interviews with respondents, complainants and witnesses during a research misconduct investigation, changing the timeline for completion of the investigation phase from 120 to 180 days,
new language for the procedures the institutional deciding official must follow when making their final determination of research misconduct findings
To see all the changes made to 42 CFR §93, an online a “redlined” version of 42 CFR §93 is available on the Ropes & Gray website.